Table of Contents
Cardiac Glycosides Overview
Cardiac glycosides more specifically digoxin is the first drug to treat heart failure, although it’s not the only drug. Cardiac glycosides also use for atrial fibrilization as well as atrial flutter.
Cardiac glycosides are derived from the plant “digitalis lanata”. For thousands of years, people have been aware of such plants.

Chemical composition of cardiac glycosides
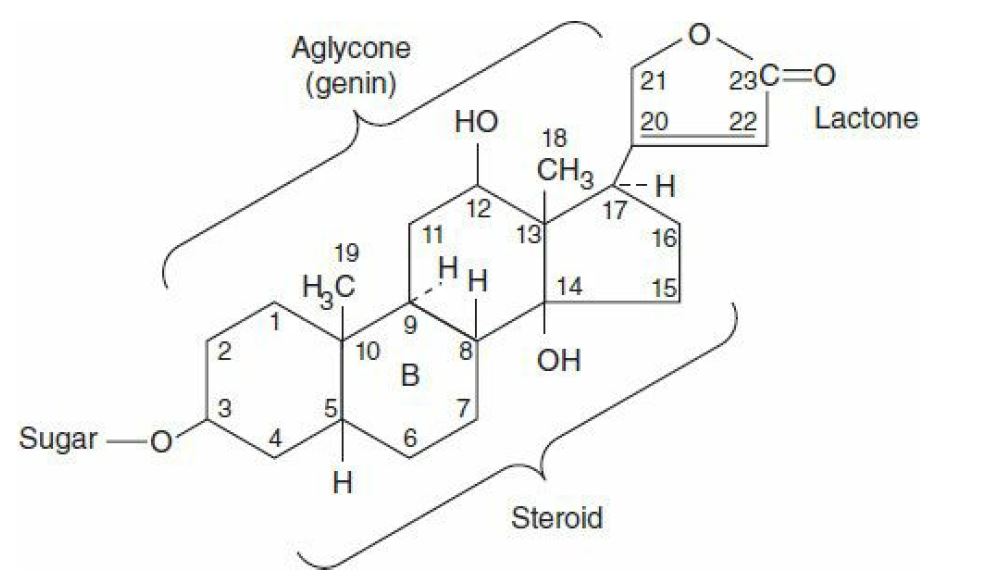
All cardiac glycosides, or cardenolides, among which digoxin is the prototype, have a specific nucleus connected to a lactone ring at the 17 positions and a sequence of sugars at the nucleus’ carbon 3. Their solubility is not pH-based since they lack a readily ionizable group.
Digoxin is derived from the white foxglove, Digitalis lanata, but many common plants (e.g., oleander, lily of the valley, milkweed, and others) provide cardiac glycosides containing similar qualities.
Pharmacokinetics of Cardiac glycosides
Digoxin, the only cardiac glycoside used in the United States, is consumed 65–80 percent after oral administration. The absorption of other glycosides ranges from 0% to nearly 100%. Both cardiac glycosides, once present bloodstream, are widely distributed to tissues, along with the central nervous system (CNS).
In humans, digoxin is not thoroughly metabolized; nearly two-thirds are excreted from the body by the kidneys. Its renal clearance is proportional to creatinine clearance, and its half-life in patients with normal renal function is 36–40 hours. Digoxin dosage can be adjusted using equations and nomograms in patients with renal disability.
Read Renal Blood Flow
Read Renal Plasma Flow
Pharmacodynamics of cardiac glycosides-How glycosides works
Digoxin has several direct and indirect cardiovascular effects, both therapeutic and toxic. Furthermore, it has negative effects on the CNS and the gut.
Almost all therapeutically useful cardiac glycosides inhibit Na+/K+-ATPase, the membrane-bound transporter known as the sodium pump. While several isoforms of this ATPase exist, each with different sensitivity to cardiac glycosides, they have been highly conserved throughout evolution. This transporter’s inhibition has been widely reported in all tissues examined across the majority of the dose range. This inhibitory activity is most likely responsible for the therapeutic impact (positive inotropy) as well as a significant portion of digitalis toxicity. Other molecular-level effects of digitalis in the heart have been observed and are discussed further below.
Since a receptor for cardiac glycosides exists on the sodium pump, some researchers believe that an endogenous digitalis-like, probably ouabain or marinobufagenin, must exist. Furthermore, apoptosis, cell growth and differentiation, immunity, and carbohydrate metabolism have been proposed as additional functions of Na+/K+-ATPase. Clinical trials suggesting some protective role of digoxin antibodies in preeclampsia have provided indirect evidence for such endogenous digitalis-like behavior.
Cardiac Effects
Mechanical effect on heart
During systole, cardiac glycosides stimulate cardiac sarcomere contraction by increasing the level of free calcium in the vicinity of contractile proteins.
The rise in calcium concentration is the consequence of a two-step process:
First, a rise in intracellular sodium concentration due to Na+/K+-ATPase inhibition(restriction);
And second, a relative decrease in calcium expulsion from the cell by the sodium-calcium exchanger. SERCA in the SR sequesters the increased cytoplasmic calcium for later release. Other methods have been suggested, but they have received little support.
The overall effect of therapeutic concentrations of a cardiac glycoside is an improvement in cardiac contractility. The rate of tension growth and relaxation is accelerated in isolated myocardial preparations, with hardly any improvement in time to peak tension. This phenomenon occurs in both normal and failing myocardium, but in the healthy individual, cardiovascular reflexes and the pathophysiology of heart failure modify the responses.
Electrical effect on heart
Digitalis effects on the electrical properties of the heart are a combination of direct and autonomic behavior.
Direct actions on cardiac cell membranes have a well-defined progression: an early, brief prolongation of the action potential, followed by a shortening (especially the plateau phase). The reduction in action potential period is most likely due to increased potassium conductance caused by increased intracellular calcium. In the absence of direct toxicity, both of these effects can be detected at therapeutic concentrations.
Effect of cardiac glycosides(digoxin) in cardiac tissue;
| Tissue/Variables | Therapeutic Dosage Effects | Toxic Dosages Effects |
| Sinus Node | Rate ↓ | Rate ↓ |
| Atrial Muscle | Refractory Period ↓ | Refractory Period & Arrhythmias ↓ |
| Atrioventricular Node | Refractory Period ↑ ; Conduction Velocity ↓ | Refractory Period & Arrhythmias ↓ |
| Purkinje Fiver, Ventricular Muscle | Refractory Period ↓ (slight) | Tachycardia, Fibrillation, Extra-systoles |
| Electrocardiogram | PR Interval ↑ ; QT Interval ↓ | Tachycardia, Fibrillation, Cardiac arrest at extremely high dosage |
Resting membrane potential is lowered (made less negative) at higher concentrations due to sodium pump suppression and decreased intracellular potassium. Following typically evoked action potentials, oscillatory depolarizing afterpotentials emerge as toxicity advances.
Afterpotentials (also known as delayed after-depolarizations, DADs) are associated with intracellular calcium pool overloading and oscillations in the free intracellular calcium ion level. When afterpotentials exceed a threshold, they cause action potentials (premature depolarizations, ectopic “beats“) to be triggered, which are coupled to the preceding normal action potentials. If afterpotentials in the Purkinje conducting system cross the threshold in this manner regularly, bigeminy will be reported on the electrocardiogram. With more intoxication, each afterpotential-evoked action potential can induce a suprathreshold afterpotential, resulting in a self-sustaining tachycardia. If left untreated, such tachycardia will progress to fibrillation; in the case of ventricular fibrillation, the arrhythmia is life-threatening unless it’s corrected.
The parasympathetic and sympathetic nervous systems are also involved in the autonomic activities of cardiac glycosides on the heart. Cardio-selective parasympathomimetic symptoms predominate at low clinical doses. Indeed, these atropine block-able effects account for a large portion of digitalis’s early electrical effects. This strategy involves baroreceptor sensitization, central vagal activation, and facilitation of muscarinic transmission at the cardiac muscle. Since cholinergic innervation is far more abundant in the atria, these acts have a greater impact on atrial and atrioventricular nodal activity than on Purkinje or ventricular function. Any of the cholinomimetic effects can be used to manage such arrhythmias. Digitalis increases sympathetic outflow at toxic levels.
This impact is not needed for normal digitalis toxicity, but it sensitizes your heart muscle and exaggerates all of the drug’s toxic effects.
Common digitalis(cardiac glycosides) side effects at a toxic dosage
- Atrioventricular junctional rhythm
- Ventricular tachycardia
- Atrioventricular blockade(2nd degree)
it is said that digitalis is capable of causing almost any arrhythmia.
Glycosides effect on other organs
Cardiac glycosides affect all excitable tissues, including smooth muscle and the central nervous system. Even outside the heart, the most common site of digitalis toxicity in the gastrointestinal tract. Anorexia, nausea, vomiting, and diarrhea are among the side effects. This toxicity is caused in part by gastrointestinal effects and in part by CNS behavior.
Vagal and chemoreceptor stimulus zone stimulation are examples of CNS effects. Disorientation, hallucinations, and blurry vision are noted less frequently, especially in the elderly. Color perception distortions can occur as a result of the above effect. Gynecomastia is a rare side effect of digitalis in males.
Read Venous Return
Chemical Interactions with Ions
Potassium and digitalis have two interactions. For starters, they inhibit each other’s binding to Na+/K+-ATPase; therefore, hyperkalemia decreases the enzyme-inhibiting effects of cardiac glycosides, while hypokalemia facilitates these effects. Second, hyperkalemia inhibits irregular cardiac automaticity. As a result, moderately increased extracellular K+ decreases the toxic effects of digitalis.
Calcium ion promotes the toxic activities of cardiac glycosides by hastening the overloading of intracellular calcium stores, which tends to be the cause of digitalis-induced irregular automaticity. As a result, hypercalcemia raises the risk of a digitalis-induced arrhythmia. The effects of magnesium ion are opposed to those of calcium ion. These interactions necessitate close monitoring of serum electrolytes in patients suffering from digitalis-induced arrhythmias.

Read Cardiac Index
Few Last Words about Cardiac Glycosides
Since cardiac glycosides have an exceedingly narrow therapeutic index and do not reduce mortality in chronic heart failure, significant efforts are being made to find better positive inotropic drugs.
Last Updated on February 23, 2022 by Learn From Doctor Team