Table of Contents
Renal Blood Flow
Renal Blood Flow: The pressure gradient through the renal vasculature (the difference between the renal artery and renal vein hydrostatic pressures) is divided by total renal vascular resistance to assess renal blood flow.
Overview of Renal Blood Flow
The combined blood flow through both kidneys in a 70-kilogram adult man is around 1100 ml/min, or about 22% of cardiac output. Considering that both kidneys account for just around 0.4 percent of total body weight, it’s easy to see why they have such a high blood supply relative to other organs. Blood flow provides nutrients to the kidneys and eliminates waste materials, much as it does with other tissues. The high flow to the kidneys, on the other hand, much exceeds this requirement. This extra flow is needed to provide enough plasma for the high rates of glomerular filtration required for precise control of body fluid volumes and solute concentrations. The processes that control renal blood flow are intimately linked to the control of GFR and the excretory functions of the kidneys, as one would assume.
Read Renal Plasma Flow
Relation of Renal Blood Flow with Oxygen Consumption
The kidneys usually use half as much oxygen as the brain per gram of body weight but have almost 7X(7 times) the blood supply. As a result, the oxygen supplied to the kidneys greatly exceeds their metabolic requirements, and arterial-venous oxygen extraction is inadequate in comparison to most other tissues.
The high rate of active sodium reabsorption by the renal tubules accounts for a significant portion of the oxygen utilized by the kidneys. Less sodium is reabsorbed and less oxygen is used as renal blood flow and GFR are decreased and less sodium is filtered. As a result, renal oxygen utilization varies with renal tubular sodium reabsorption, which is linked to GFR and sodium filtration rate. Renal sodium reabsorption also stops when glomerular filtration stops, and oxygen utilization drops to around one-fourth of normal. The renal cells’ essential metabolic needs are reflected in this residual oxygen consumption(utilization).
Determinants(factors) of Renal Blood Flow
(Renal artery pressure – Renal venous pressure) / Total renal vascular rresistance
In certain circumstances, renal artery pressure is about equal to systemic arterial pressure, and renal vein pressure is about 3 to 4 mm Hg. The number of resistances in the individual vasculature segments, including the arteries, arterioles, capillaries, and veins, determines the overall vascular resistance in the kidneys, as it does in other vascular beds.
The interlobular arteries, afferent arterioles, and efferent arterioles account for the majority of renal vascular resistance. The sympathetic nervous system, various hormones, and local internal renal control mechanisms control the resistance of these vessels. If renal artery and renal vein pressures stay unchanged, a rise in the resistance of either of the vascular segments of the kidneys diminishes renal blood flow, while a decrease in vascular resistance leads to increased renal blood flow.
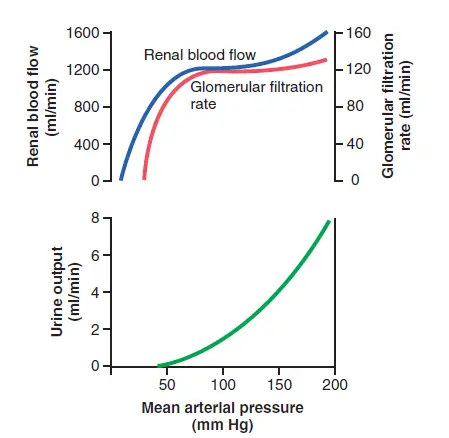
While changes in arterial pressure have an impact on renal blood flow, the kidneys have effective mechanisms for maintaining renal blood flow and GFR in a relatively constant range between 80 and 170 mm Hg, a process known as autoregulation. This ability to self-regulate is achieved by processes that are unique to the kidneys.
Read Renal Infarction.
The Physiological Control Mechanism of RBF and GFR
Including glomerular hydrostatic pressure and the glomerular colloid, the most complex and physiologically regulated GFR determinant
The pressure of the osmotic. The sympathetic nervous system, the hormones and autacoids (vasoactive substances released into the kidneys and acting locally), and other feedback controls inherent in the kidneys, in turn, affect these variables. these variables
Effect of the Sympathetic Nervous System to Maintain the RBF and GFR
Sympathetic nerve fibers are richly internalized by all the blood vessels of the kidneys, including the afferent and the effectual arterioles Strong renal sympathetic activation can reduce kidney blood and GFR blood flow and can reduce the arteries in the kidney. Moderate or mild stimulus does not affect the blood supply of the renal blood and GFR. As mentioned in the study, however, even mild increases in renal sympathy can cause a decrease in sodium and excretion of water through increased renal tubular reabsorption.
In severely intense, GFR-related disturbances that last for several minutes to several hours, such as those caused by the body defense mechanism, brain ischemia, or severe blood pressure, the renal sympathetic nerves appear to be most significant. A sympathetic tone seems to have little effect on renal blood circulation in the healthy resting individual.
Read Arterial Bleeding
Hormonal Control of Renal Blood Flow
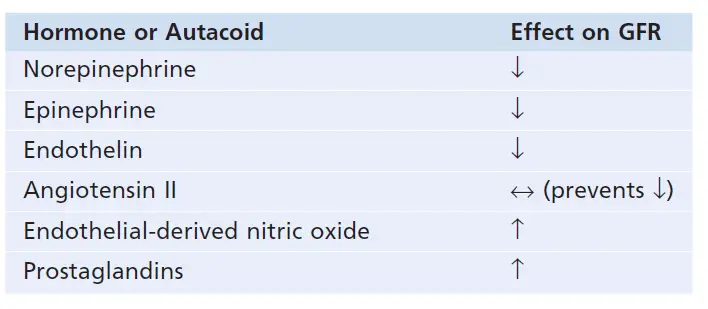
Constrict the renal blood vessels and decrease GFR: norepinephrine, epinephrine, and endothelin
Norepinephrine and epinephrine emitted from the adrenal medulla include hormones that constrict afferent and efferent arterials causing reductions in GFR and renal blood flow. Generally speaking, these blood hormones have no impact on renal hemodynamics, except under extreme circumstances like serious hemorrhage, since they have a parallel function of the sympathetic nervous system.
The peptide that may be released from the weakened vascular endothelial cells of the kidneys or other tissues is an endothelin. It is not fully understood the physiological function of this autacoid. But, when a blood vessel is severed which damages the endothelium, endothelin will contribute to hemostasis (minimizing the blood loss) and release this powerful blood constrictor. In several disease states associated with vascular injury, including pregnancy toxemia, acute renal failure, and chronic uremia, plasma endothelin levels have also been increased and some of these pathophysiological conditions may contribute to renal vasoconstriction and decreased GFR.
In most physiological conditions, angiotensin II preferentially restricts efferent arterioles.
In certain circumstances, renal artery pressure is about equal to systemic arterial pressure, and renal vein pressure is about 3 to 4 mm Hg. The number of resistances in the individual vasculature segments, including the arteries, arterioles, capillaries, and veins, determines the overall vascular resistance in the kidneys, as it does in other vascular beds.
The interlobular arteries, afferent arterioles, and efferent arterioles account for the majority of renal vascular resistance. The sympathetic nervous system, various hormones, and local internal renal control mechanisms control the resistance of these vessels. If renal artery and renal vein pressures stay unchanged, a rise in the resistance of either of the vascular segments of the kidneys diminishes renal blood flow, while a decrease in vascular resistance leads to increased renal blood flow.
While changes in arterial pressure have an impact on renal blood flow, the kidneys have effective mechanisms for maintaining renal blood flow and GFR in a relatively constant range between 80 and 170 mm Hg, a process known as autoregulation. This ability to self-regulate is achieved by processes that are intrinsic to the kidneys.
Read Pulmonary Vascular Resistance
Endothelial-derived nitric oxide reduces the resistance to kidney vascular and enhances GFR.
Endothelial-derived nitric oxide is an autacoid that lowers renal vascular resistance and is released in the body by the vascular endothelium. Since it helps the kidneys to excrete normal quantities of sodium and water, a low level of nitric oxide production appears to be necessary for maintaining renal vasodilation. As a result, medications that prevent nitric oxide production increase renal vascular resistance and decrease GFR and urinary sodium excretion, resulting in high blood pressure. Damage to the vascular endothelium and impaired nitric oxide output can lead to increased renal vasoconstriction and elevated blood pressure in some hypertensive and atherosclerotic patients.
The decreased renal vascular resistance occurs by prostaglandins and bradykinin and tends to raise the GFR.
The prostaglandins (PGE2 and PGI2), as well as bradykinin, are hormones and autacoids that trigger vasodilation and increased renal blood flow and GFR. Although these vasodilators do not appear to play a significant role in controlling renal blood flow or GFR in normal circumstances, they can reduce the renal vasoconstrictor effects of sympathetic nerves or angiotensin II, especially their effects on afferent arterioles. The prostaglandins can help prevent excessive reductions in GFR and renal blood flow by opposing afferent arteriole vasoconstriction. NSAIDs, such as aspirin, that inhibit prostaglandin synthesis, can cause significant reductions in GFR under extreme situations, such as volume depletion or after surgery.
Read Renal Parenchymal Disease.
Autoregulation of RBF and GFR
Despite significant increases in arterial blood pressure, the kidneys’ internal feedback systems usually keep renal blood flow and GFR relatively stable. These processes continue to work in blood-perfused kidneys that have been separated from the body, even though they are no longer connected to the rest of the body. Autoregulation refers to the relative consistency of GFR and renal blood flow.
Despite changes in arterial pressure, the main role of blood flow autoregulation in most tissues other than the kidneys is to keep constant oxygen and nutrient supply and to eliminate metabolic waste products. The normal blood circulation in the kidneys is much higher than what is expected for these functions. Autoregulation in the kidneys is primarily responsible for maintaining a relatively constant GFR and allowing precise control of renal water and solvent excretion.
Despite significant arterial pressure variations that occur during a person’s daily activities, the GFR is usually autoregulated (that is, it remains fairly constant). A decrease in arterial pressure to as low as 70 to 75 mm Hg or an increase to as high as 160 to 180 mm Hg, for example, typically results in a GFR shift of less than 10%. Renal blood flow is autoregulated in general in coordination with GFR, but GFR is more effectively autoregulated in such circumstances.
Read Renal Fascia
Importance of Autoregulation of GFR
Although the renal autoregulatory mechanisms are not ideal, they do help to avoid potentially large increases in GFR and renal excretion of fluid and solutes that would occur if blood pressure changed. Consider the relative magnitudes of glomerular filtration, tubular reabsorption, and renal excretion, as well as the improvements in renal excretion that would occur without autoregulatory processes to understand the quantitative value of autoregulation.
GFR is normally about 180 L/day, with tubular reabsorption at 178.5 L/day, leaving 1.5 L/day for urinary excretion. Without autoregulation, a modest rise in blood pressure (from 100 to 125 mm Hg) will result in a similar 25% increase in GFR (from 180 to 225 L/day). The urine flow would increase to 46.5 L/day (the difference between GFR and tubular reabsorption) if tubular reabsorption remained stable at 178.5 L/day, resulting in a cumulative increase in the urine of more than 30-fold. Since total plasma volume is only around 3 liters, a shift like this will rapidly deplete blood volume.
Changes in arterial pressure have a much smaller impact on urine volume for two reasons:
- Renal autoregulation prevents major changes in GFR.
- The renal tubules have additional adaptive mechanisms that allow them to increase their reabsorption rate as GFR rises, a phenomenon known as tubuloglomerular balance.
Also with these special control mechanisms in place, changes in arterial pressure still have a direct impact on renal fluid and sodium excretion; this is known as pressure diuresis or pressure natriuresis, and it is critical for maintaining body fluid volumes and arterial pressure.
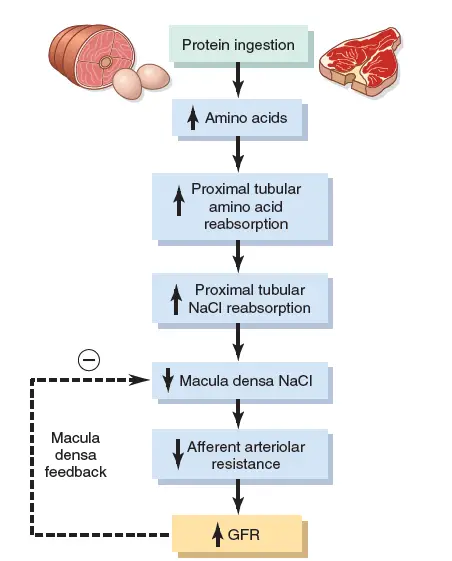
Effect of Tubuloglomerular Feedback Mechanism and Autoregulation of GFR and RBF
The kidneys have a feedback system linked to changes in the concentration of sodium chloride in the macula densa with control of renal arterial resistance and GFR auto-regulation. This feedback ensures that sodium chloride is delivered to the distal tube relatively constantly and helps avoid renal excretion fluctuations which otherwise would happen. This feedback autoregulates, in certain cases, parallel renal blood flow and GFR. As this mechanism is primarily geared to stabilizing the supply of sodium chloride to the distal tubule, however, instances are occurring when GFR is controlled at the expense of renal blood flow changes, as discussed below. In other cases, GFR may potentially be changed as a result of primary improvements in sodium reabsorption of the reinforced renal tubular chloride.
The tubuloglomerular feedback mechanism contains two components that work together to control GFR:
- A strong mechanism for afferent arteriolar.
- A powerful mechanism for efferent arteriolar.
These feedback mechanisms rely on special juxtaglomerular complex anatomical arrangements.
The juxtaglomerular complex includes macula densa cells in the first part of the distal tubule in the afferent and efferent arterioles. The macula densa is a special epithelial cell group in the distal tubules which come into close contact with afferent arterioles. The macula densa cells have Golgi apparatus, which are intracellular, secretory organs targeted at the arterioles, which means these cells can secrete a substance in the arterioles.
Myogenic Autoregulation of RBF(Renal Blood Flow)
The capacity of individual blood vessels in order to avoid stretching under increased arterial pressure, a phenomenon called the myogenic mechanism, is another mechanism contributing to maintaining a relatively constant renal blood flow and GFR. Studies of the individual blood vessels in the body (especially small arterioles) have shown that the vascular smooth muscle contraction is responding to the increased tension on the wall or the wall. Extending the vascular wall enables the extracellular fluid to transfer more calcium ions into the cells, thereby contracting them. This contraction protects the artery from excessive stretching while also preventing excessive increases in the renal blood flow and GFR as arterial pressure increases by increasing vascular resistance.
While the myogenic process possibly functions in most arterioles in the whole body, some physiologists have called into question its significance in the flow of renal blood and GFR autoregulation because it does not specifically detect changes in renal blood or GFR per se. This is a pressure-sensitive mechanism. This mechanism, on the other hand, maybe necessary to protect the renal against hypertension-induced injury. As the blood pressure rises suddenly, myogenic blood pressure reaction in afferent arterioles takes seconds and thus reduces transmission to the glomerular capillary arteries from elevated arterial pressure.
Few Last Words
Renal blood flow is 22% of total body blood volume. This high volume is required to keep our blood clean and healthy. RBF depends on certain situations and is regulated via hormonal, neurogenic, myogenic, and other mechanisms.
Last Updated on February 23, 2022 by Learn From Doctor Team