Table of Contents
Pulmonary Vascular Resistance Overview
Pulmonary vascular resistance: The mean pulmonary arterial pressure in the pulmonary system is 16 mm Hg, and the mean left atrial pressure is 2 mm Hg, for a total pressure difference of 14 mm. As a result, when cardiac output is normal at around 100 ml/sec, total pulmonary vascular resistance is calculated to be around 0.14 PRU (about one-seventh that in the systemic circulation).
Widely Used Formula For Calculating Pulmonary Vascular Resistance

To understand pulmonary vascular resistance properly we have to understand the physiology of resistance to blood flow first.
Read Renal Blood Flow
Physiology of Resistance To Blood Flow
Resistance unit
Resistance impedes blood flow in a vessel that cannot be measured directly. Instead, resistance must be measured using blood flow and pressure differential measures between two points in the vessel. If the pressure difference between two points is 1 mm Hg and the flow rate is 1 ml/sec, the resistance is one peripheral resistance unit, abbreviated PRU.
Resistance is CGS(centimeter, gram, or seconds) units
To convey resistance, a simple physical unit known as the CGS unit is often used. This is a dyne sec/cm5 unit. The following formula can be used to measure resistance in these units:
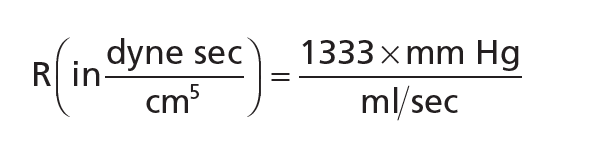
Relation Between Total Vascular Resistance and Pulmonary Vascular Resistance
The rate of blood flow through the complete circulatory system is equal to the amount of blood pumping by the heart, or the cardiac output. This is about 100 ml/sec in an adult human individual. The pressure difference between the systemic arteries and systemic veins is approximately 100 mm Hg. As a result, the average peripheral resistance (PRU) of the complete systemic circulation is approximately 100/100, or 1 PRU.
Systemic Vascular Resistance(SVR) Calculation

Total peripheral resistance will sometimes reach 4 PRU in situations where all blood vessels in the body become severely constricted.
When the vessels dilate significantly, the resistance will drop to as low as 0.2 PRU.
The mean pulmonary arterial pressure in the pulmonary system is 16 mm Hg, and the mean left atrial pressure is 2 mm Hg, for a total pressure difference of 14 mm. As a result, when cardiac output is natural at around 100 ml/sec, full pulmonary vascular resistance is calculated to be around 0.14 PRU (roughly one-seventh of that in the systemic circulation).
Read Renal Plasma Flow
Conductance of Blood Vessels
The conductance of a vessel, pipe, or channel is a measurement of the blood flow through it for a given pressure difference. This calculation is commonly expressed in milliliters per second per millimeter of mercury pressure, but it can also be measured in liters per second per millimeter of mercury pressure or any other unit of blood pressure and flow.
Conductance is reciprocal of the resistance.
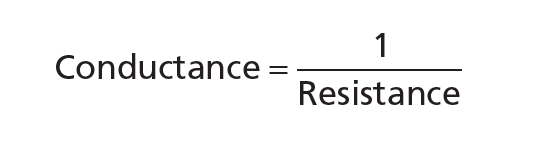
Minor changes in vessel diameter have a major impact on its conductance.
Conductance ∞ Diameter4
Effect of blood hematocrit and viscosity in pulmonary vascular resistance
It is worth remembering that the viscosity of the blood is also a significant factor. If all other variables remain constant, the higher the viscosity, the lower the flow in a vessel.
Besides that, the viscosity of normal blood is about 3 times that of water. So, a question may arise, what causes the blood to be so viscous (thick). It is primarily due to a large number of suspended red cells in the blood, each of which exerts a frictional pull against neighboring cells and the blood vessel surface.
If an individual has a hematocrit of 40, it means that cells make up 40% of the blood volume and plasma makes up the rest. Adult men have a hematocrit of about 42, while women have a hematocrit of about 38. These values differ greatly depending on whether or not the individual has anemia, the level of physical activity, and the altitude at which the person lives.
Blood is centrifuged in a calibrated tube to assess hematocrit. The calibration enables direct observation of the cell percentage.
Effect of Pressure in Pulmonary Vascular Resistance
One might think a rise in arterial pressure results in a proportionate increase in blood flow through the body’s various tissues. However, the impact of arterial pressure on blood circulation in many tissues is normally much less than one would imagine.
This is because a rise in arterial pressure not only raises the force that forces blood through the arteries but also causes compensatory increases in vascular resistance within a few seconds by activating the local control mechanisms. In contrast, as arterial pressure is decreased, vascular resistance is immediately reduced in most tissues and blood flow is maintained at a steady rate. Blood flow autoregulation refers to a tissue’s ability to adjust its vascular resistance and maintain regular blood flow during increases in arterial pressure between 70 and 175 mm Hg.
Role of Pulmonary Vascular Resistance at Respiration at Birth
Due to the patent ductus arteriosus and foramen ovale, the left and right hearts pump in parallel instead of sequence in the fetus. The circulation must transition from a parallel to a sequential scheme, as well as from placental to pulmonary gas exchange, at birth. This must be done in large part within moments of birth, though full adjustments can take many hours. The fall in pulmonary vascular resistance that occurs when the lungs are first filled with gas is critical in initiating the changes. After the lungs are enlarged by the first few breaths, the pulmonary vascular resistance drops to less than 20% of what it was in utero.
The stimulation that causes the baby to begin breathing after delivery is not completely known. There is no indication that hypoxia plays a major role in normal labor, and it is more likely that the most relevant causes are sudden exposure to light, sound, cold air, and other physical stimuli. Oxygen is a potent vasodilator of the pulmonary vascular tree that works by producing nitric oxide (NO). High arterial oxygen pressure and bradykinin, which is released by the lungs during their initial inflation, are vasoconstrictors of the umbilical arteries and ductus arteriosus.
The pressure in the left atrium rises as blood returns from the lungs, closing the foramen ovale by pressing the valve that protects it against the interatrial septum. The ductus arteriosus tightens, and this, combined with a decrease in pulmonary vascular resistance, guides the majority of the right ventricle’s output into the lungs. Owing to severe intimal thickening, the ductus arteriosus is functionally closed within a few hours of birth, and permanent anatomic closure occurs within the next 24–48 hours. Furthermore, relatively high amounts of vasodilators, particularly prostaglandin F2, are available in the ductus in utero, and synthesis of this prostaglandin is inhibited by cyclooxygenase blockade at birth. Many premature infants’ ducti fail to close spontaneously, but drugs that inhibit cyclooxygenase can cause the closure. In this case, NO can also play a role in preserving ductal patency.
After pulmonary respiration is developed, extremely oxygenated blood carrying pulmonary bradykinin enters the umbilical arteries, causing constriction. Cold and handling of the cord stimulate further constriction, and the arteries are fully constricted within 3 to 5 minutes of birth. Venous flow persists as a result of the elevated placental pressure inside the contracted uterus and the newborn’s negative intrathoracic pressure (immediately 30 to 50 mm Hg). Within 5 minutes, the flow of blood within the umbilical cord stops, and there is no leftover blood within the placenta. The “placental transfusion” refers to the transfer of blood within the fetoplacental circulation following birth.
Read Tension Pneumothorax
Few Last Words
Pulmonary Vascular Resistance calculation is important in some cases to diagnose the patient’s condition and understand the circulatory system properly. PVR depends on certain factors such as; vessel condition, shunt vessels, any injury, disease or conditions, etc.
Last Updated on February 23, 2022 by Learn From Doctor Team